|
Textbook content produced by OpenStax is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. We recommend using aĪuthors: John McMurry, Professor Emeritus Use the information below to generate a citation. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, 2D IR experiments are performed with bandwidth sufficient to cover the fundamental and overtone bands of amide I’ and amide II’ using a 2D FT spectrometer that is described in detail elsewhere. The three types of amides can be distinguished from each other by infrared spectroscopy as will be seen in the next column. Then you must include on every physical page the following attribution: Amides also exhibit hydrogen bonding, resulting in somewhat broadened N-H stretching peaks. secondary amine, amide RNR-H, RCONHR 3300-3500 S(br) 20, 21 tertiary amine. If you are redistributing all or part of this book in a print format, The wavelength of light in the IR region varies. Want to cite, share, or modify this book? This book uses theĬreative Commons Attribution-NonCommercial-ShareAlike License IR will not give a two state behavior since the various intermediates will. 
C–C bond nearest the nitrogen atom is broken, yielding an alkyl radical and a resonance-stabilized, nitrogen-containing cation. This is consistent with what happens when the molecule. For example, morphine has the formula C 17H 19NO 3 and a molecular weight of 285 amu.Īlkylamines undergo a characteristic α cleavage in the mass spectrometer, similar to the cleavage observed for alcohols ( Section 17.11). The logic behind the rule derives from the fact that nitrogen is trivalent, thus requiring an odd number of hydrogen atoms. An odd-numbered molecular ion usually means that the unknown compound has one or three nitrogen atoms, and an even-numbered molecular ion usually means that a compound has either zero or two nitrogen atoms. 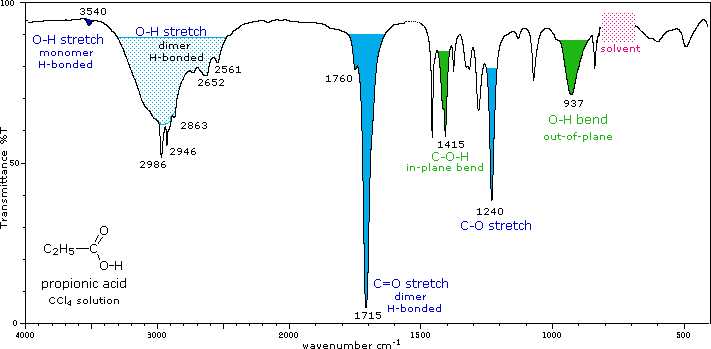 
Thus, the presence of nitrogen in a molecule is detected simply by observing its mass spectrum. 1 2 In physical and analytical chemistry, infrared spectroscopy (IR spectroscopy) is a technique used to identify. The nitrogen rule of mass spectrometry says that a compound with an odd number of nitrogen atoms has an odd-numbered molecular weight. An infrared spectroscopy correlation table (or table of infrared absorption frequencies) is a list of absorption peaks and frequencies, typically reported in wavenumber, for common types of molecular bonds and functional groups. What are the structures of A and B? Mass Spectrometry IR spectra result from the convolution (see Chapter A3) of the sample. Considerable information is available on the infrared spectra of amides. The IR and 1H NMR spectra of B are shown. Amide I CH3 CH3 Amide II Amide III e c n a b r o s b A O A H C N CH3 CH3. Compound A, C 6H 12O, has an IR absorption at 1715 cm –1 and gives compound B, C 6H 15N, when treated with ammonia and NaBH 4.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
